
Measurements at the Mauna Loa Observatory stopped after the 2022 eruption of the Mauna Loa volcano, when lava flow crossed the access road and took out power lines to the facility. The observatory remains inaccessible by vehicle and without power from the local utility company.
Observatory staff has established limited solar power in four observatory buildings and restored approximately 33 percent of the measurements onsite, including the Global Monitoring Laboratory and Scripps critical CO2 records and other atmospheric measurements.
Media can contact: Theo Stein (303) 819-7409 (theo.stein@noaa.gov) or Karin Vergoth 303-632-6413 (karin.vergoth@noaa.gov)
 National Oceanic and Atmospheric Administration (NOAA),
National Oceanic and Atmospheric Administration (NOAA),
Earth System Research Laboratory (ESRL)
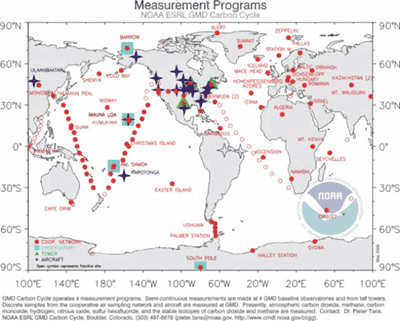
The NOAA CMDL CCGG cooperative global air sampling network is an international effort, and includes regular discrete air flask samples from both Mauna Loa Observatory and Cape Kumukahi, Hawaii. Complete information about the program is available at http://gml.noaa.gov/ccgg/flask.html.
The program measures the following (where ppm is parts-per-million, ppb is parts-per-billion, ppt is parts-per-trillion, pm is per-million, and per mil is per-thousand):
| Measurement | Chemical Formula | Units |
|---|---|---|
| Carbon Dioxide | CO2 | ppm |
| Methane | CH4 | ppb |
| Carbon Monoxide | CO | ppb |
| Hydrogen | H 2 | ppb |
| Nitrous Oxide | N2O | ppb |
| Sulfur Hexaflouride | SF6 | ppt |
| isotopic ratio of carbon dioxide | Carbon-13 / Carbon-12 | per mil |
| isotopic ratio of carbon dioxide | Oxygen-18 / Oxygen-16 | per mil |
Flask samples are taken at Mauna Loa Observatory and Cape Kumukahi, Hawaii, and are sent to the NOAA ESRL GMD laboratory in Boulder, Colorado. They are then analyzed with the following techniques:
In order to determine temporal and spatial variations in the global atmosphere, deduce better estimates of sources and sinks, and better understand the carbon cycle.
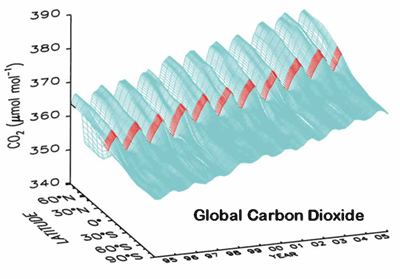
Carbon dioxide is increasing by ~1.5 parts per million per year;
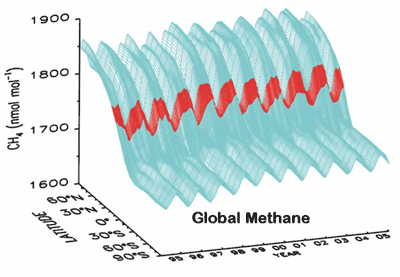
Methane was increasing, but has been nearly constant for the past 2 years;
Nitrous oxide and sulphur hexaflouride are increasing. Sulfur hexaflouride (SF6) is the most potent greenhouse gas on a molecule-to-molecule basis, and has an atmospheric lifetime of about 3,000 years! But, since SF6 concentrations are relatively low at present, their greenhouse effet will not be felt for many years. SF6 is used in the electrical power transmission industry and its sources are mainly in the northern hemisphere.
Carbon-13 / Carbon-12 is decreasing.
The CCGG flask sample network consists of the sites shown on the map below (click map to enlarge).
Carbon dioxide is the most important anthropogenic “greenhouse gas” (it produces about 60% of present day greenhouse gas climate forcing), followed by methane (about 20%). Nitrous oxide and sulphur hexaflouride are also infrared absorbers. Carbon monoxide affects the greenhouse gases through its atmospheric chemistry. A better understanding of the carbon cycle is crucial to make informed decisions about future climate and energy policies.
More information, plus many figures, are available from the CCGG web site.
Pieter Tans
303-497-6678
Ed Dlugokencky
303-497-6228
Aidan Colton
808-933-6965 (x233)
Paul Fukumura
808-933-6965 (x223)
http://www.esrl.noaa.gov/
gmd/ccgg/index.html
08/20/1969
In Situ Carbon Monoxide
In Situ Carbon Dioxide
In Situ Methane
Greenhouse Gases