
Chlorofluorocarbons (CFCs)
(published in The Chapman & Hall Encyclopedia of Environmental Science, edited by David E. Alexander and Rhodes W. Fairbridge, pp pp.78-80, Kluwer Academic, Boston, MA, 1999.)
Chlorofluorocarbons (CFCs) are nontoxic, nonflammable chemicals containing atoms of carbon, chlorine, and fluorine. They are used in the manufacture of aerosol sprays, blowing agents for foams and packing materials, as solvents, and as refrigerants. CFCs are classified as halocarbons, a class of compounds that contain atoms of carbon and halogen atoms. Individual CFC molecules are labeled with a unique numbering system. For example, the CFC number of 11 indicates the number of atoms of carbon, hydrogen, fluorine, and chlorine (e.g. CCl3F as CFC-11). The best way to remember the system is the "rule of 90" or add 90 to the CFC number where the first digit is the number of carbon atoms (C), the second digit is the number of hydrogen atoms (H), and the third digit is number of the fluorine atoms (F). The total number of chlorine atoms (Cl) are calculated by the expression: Cl = 2(C+1) - H - F. In the example CFC-11 has one carbon, no hydrogen, one fluorine, and therefore 3 chlorine atoms.
Refrigerators in the late 1800s and early 1900s used the toxic gases, ammonia (NH3), methyl chloride (CH3Cl), and sulfur dioxide (SO2), as refrigerants. After a series of fatal accidents in the 1920s when methyl chloride leaked out of refrigerators, a search for a less toxic replacement begun as a collaborative effort of three American corporations- Frigidaire, General Motors, and Du Pont. CFCs were first synthesized in 1928 by Thomas Midgley, Jr. of General Motors, as safer chemicals for refrigerators used in large commercial appilications1. Frigidaire was issued the first patent, number 1,886,339, for the formula for CFCs on December 31, 1928. In 1930, General Motors and Du Pont formed the Kinetic Chemical Company to produce Freon (a Du Pont tradename for CFCs) in large quantities. By 1935 Frigidaire and its competitors had sold 8 million new refrigerators in the United States using Freon-12 (CFC-12) made by the Kinetic Chemical Company and those companies that were licensed to manufacture this compound. In 1932 the Carrier Engineering Corporation used Freon-11 (CFC-11) in the worldís first self-contained home air-conditioning unit, called the "Atmospheric Cabinet".; Because of the CFC safety record for nontoxicity, Freon became the preferred coolant in large air-conditioning systems. Public health codes in many American cities were revised to designate Freon as the only coolant that could be used in public buildings. After World War II, CFCs were used as propellants for bug sprays, paints, hair conditioners, and other health care products. During the late 1950s and early 1960s the CFCs made possible an inexpensive solution to the desire for air conditioning in many automobiles, homes, and office buildings. Later, the growth in CFC use took off worldwide with peak, annual sales of about a billion dollars (U.S.) and more than one million metric tons of CFCs produced.
Whereas CFCs are safe to use in most applications and are inert in the lower atmosphere, they do undergo significant reaction in the upper atmosphere or stratosphere. In 1974, two University of California chemists, Professor F. Sherwood Rowland and Dr. Mario Molina, showed that the CFCs could be a major source of inorganic chlorine in the stratosphere following their photolytic decomposition by UV radiation. In addition, some of the released chlorine would become active in destroying ozone in the stratosphere2. Ozone is a trace gas located primarily in the stratosphere (see ozone). Ozone absorbs harmful ultraviolet radiation in the wavelengths between 280 and 320 nm of the UV-B band which can cause biological damage in plants and animals. A loss of stratospheric ozone results in more harmful UV-B radiation reaching the Earth's surface. Chlorine released from CFCs destroys ozone in catalytic reactions where 100,000 molecules of ozone can be destroyed per chlorine atom.
A large springtime depletion of stratospheric ozone was getting worse each following year. This ozone loss was described in 1985 by British researcher Joe Farman and his colleagues3. It was called ìthe Antarctic ozone holeî by others. The ozone hole was different than ozone loss in the midlatitudes. The loss was greater over Antarctic than the midlatitudes because of many factors: the unusually cold temperatures of the region, the dynamic isolation of this ìholeî, and the synergistic reactions of chlorine and bromine4. Ozone loss also is enhanced in polar regions as a result of reactions involving polar stratospheric clouds (PSCs)5 and in midlatitudes following volcanic eruptions. The need for controlling the CFCs became urgent.
In 1987, 27 nations signed a global environmental treaty, the Montreal Protocol to Reduce Substances that Deplete the Ozone Layer6, that had a provision to reduce 1986 production levels of these compounds by 50% before the year 2000. This international agreement included restrictions on production of CFC-11, -12, -113, -114, -115, and the Halons (chemicals used as a fire extinguishing agents). An amendment approved in London in 1990 was more forceful and called for the elimination of production by the year 2000. The chlorinated solvents, methyl chloroform (CH3CCl3), and carbon tetrachloride (CCl4) were added to the London Amendment.
Large amounts of reactive stratospheric chlorine in the form of chlorine monoxide (ClO) that could only result from the destruction of ozone by the CFCs in the stratosphere were observed by instruments onboard the NASA ER-2 aircraft and UARS (Upper Atmospheric Research Satellite) over some regions in North America during the winter of 19927,8. The environmental concern for CFCs follows from their long atmospheric lifetime (55 years for CFC-11 and 140 years for CFC-12, CCl2F2)9 which limits our ability to reduce their abundance in the atmosphere and associated future ozone loss. This resulted in the Copenhagen Amendment that further limited production and was approved later in 1992. The manufacture of these chemicals ended for the most part on January 1, 1996. The only exceptions approved were for production within developing countries and for some exempted applications in medicine (i.e., asthma inhalators) and research. The Montreal Protocol included enforcement provisions by applying economic and trade penalties should a signatory country trade or produce these banned chemicals. A total of 148 signatory countries have now signed the Montreal Protocol. Atmospheric measurements CFC-11 and CFC-12 reported in 1993 showed that their growth rates were decreasing as result of both voluntary and mandated reductions in emissions9. Many CFCs and selected chlorinated solvents have either leveled off (Figure 1) or decreased in concentration by 19949,10.
The demand for the CFCs was accomodated by recycling, and reuse of existing stocks of CFCs and by the use of substitutes. Some applications, for example degreasing of metals and cleaning solvents for circuit boards, that once used CFCs now use halocarbon-free fluids, water (sometimes as steam), and diluted citric acids. Industry developed two classes of halocarbon substitutes- the hydrochlorofluorocarbons (HCFCs) and the hydrofluorocarbons (HFCs). The HCFCs include hydrogen atoms in addition to chlorine, fluorine, and carbon atoms. The advantage of using HCFCs is that the hydrogen reacts with tropospheric hydroxyl (OH), resulting in a shorter atmospheric lifetime. HCFC-22 (CHClF2) has an atmospheric lifetime of about 13 years11 and has been used in low-demand home air-conditioning and some refrigeration applications since 1975. However, HCFCs still contain chlorine which makes it possible for them to destroy ozone. The Copenhagen amendment calls for their production to be eliminated by the year 2030. The HFCs are considered one of the best substitutes for reducing stratospheric ozone loss because of their short lifetime and lack of chlorine. In the United States, HFC-134a is used in all new domestic automobile air conditioners. For example, HFC-134a is growing rapidly in 1995 at a growth rate of about 100% per year with an atmospheric lifetime of about 12 years12. (The "rule of 90" also applies for the chemical formula of HCFCs and HFCs.)
Use of the CFCs, some chlorinated solvents, and Halons should become obsolete in the next decade if the Montreal Protocol is observed by all parties and substitutes are used. The science that became the basis for the Montreal Protocol resulted in the 1995 Nobel Prize for Chemistry. The prize was awarded jointly to Professors F. S. Rowland at University of California at Irvine, M. Molina at the Massachusetts Institute of Technology, Cambridge, and Paul Crutzen at the Max-Planck-Institute for Chemistry in Mainz, Germany, for their work in atmospheric chemistry, particularly concerning the formation and decomposition of ozone (in particular, by the CFCs and oxides of nitrogen).
1Midgley, T., and Henne, A., Organic fluorides as refrigerants, Industrial and Engineering Chemistry, 22, 542-547, 1930.
2Molina, M.J., and F.S. Rowland, Stratospheric sink for chlorofluoromethanes: Chlorine atom catalyzed destruction of ozone, Nature, 249, 810-814, 1974.
3Farman, J.C., B.G. Gardiner, and J.D. Shanklin, Large losses of total ozone in Antarctica reveal seasonal ClOx/NOx interaction, Nature, 315,207-210, 1985.
4McElroy, M.B., R.J. Salawitch, S.C. Wofsy, and J.A. Logan, Reductions of Antarctic ozone due to synergistic interactions of chlorine and bromine, Nature, 321, 759-762, 1986.
5Solomon, S., R.R. Garcia, F.S. Rowland, and D.J. Wuebbles, On the depletion of Antarctic ozone, Nature, 321, 755-758, 1986.
6Montreal Protocol on Substances that Deplete the Ozone Layer, 15 pp, United Nations Environmental Programme (UNEP), New York, 1987.
7Toohey, D.W., L.M. Avallone, L.R. Lait, P.A. Newman, M.R. Schoeberl, D.W. Fahey, E.L. Woodbridge, and J.G. Anderson, The seasonal evolution of reactive chlorine in the northern hemisphere stratosphere, Science, 261, 1134-1136, 1993.
8Waters, J., L. Froidevaux, W. Read, G. Manney, L. .Elson, D. Flower, R. Jarnot, and R. Harwood, Stratospheric ClO andozone from the Microwave Limb Sounder on the Upper Atmosphere Research Satellite, Nature, 362, 597-602, 1993.
9Elkins, J.W., T.M. Thompson, T.H. Swanson, J.H. Butler, B.D. Hall, S.O. Cummings, D.A. Fisher, and A.G. Raffo, Decrease in the growth rates of atmospheric chlorofluorocarbons 11 and 12, Nature, 364 , 780-783, 1993.
10Prinn, R.G., R.F. Weiss, B.R. Miller, J. Huang, F.N. Alyea, D.M. Cunnold, P.J. Fraser, D.E. Hartley, and P.G. Simmonds, Atmospheric trends and lifetimes of CH3CCl3 and global OH concentrations, Science, 269, 187-192, 1995.
11Montzka, S.A., R.C. Myers, J.H. Butler, S.C. Cummings, and J.W. Elkins, Global tropospheric distribution and calibration scale of HCFC-22, Geophysical Research Letters, 20 (8), 703-706, 1993.
12Montzka, S.A., R.C. Myers, J.H. Butler, J.W. Elkins,
L.T. Lock, A.D. Clarke, and A.H. Goldstein, Observations of HFC-134a in the
remote troposphere, Geophysical Research Letters, 23, 169-172,
1996.
Cagin, S., and P. Dray, Between Earth and Sky: How CFCs changed our world and threatened the ozone layer, 512 pp., Pantheon Press, New York, 1993.
Scientific Assessment of Ozone Depletion: 1994, edited by D. L. Albritton, R. T. Watson, and R. J. Aucamp, 37, 451 pp., World Meteorological Organization (WMO), Geneva, 1995.
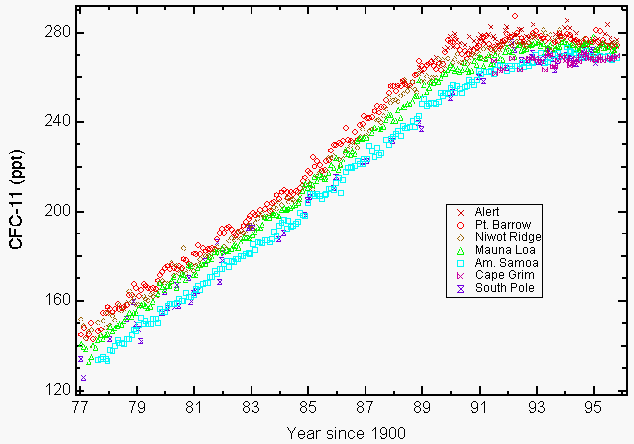
Figure 1: The accumulation of chlorofluorocarbon-11 (CFC-11) in the atmosphere levels off as a result of voluntary and mandated emission reductions. Monthly means reported as dry mixing ratios in parts per trillion (ppt) for CFC-11 at ground level for four NOAA/CMDL stations (Pt. Barrow, Alaska; Mauna Loa, Hawaii; Cape Matatula, American Samoa; and South Pole) and three cooperative stations (Alert, Northwest Territories, Canada (Atmospheric Environment Service); Niwot Ridge, Colorado (University of Colorado); Cape Grim Baseline Air Pollution Station, Tasmania, Australia, (Commonwealth Scientific and Industrial Research Organization)9. (Courtesy of NOAA/CMDL)