Research Overview
Science Introduction & Operational Plan [Science Questions]
ER-2 & DC-8 Aircraft Mission To Investigate Antarctic Ozone In Late Winter 1987
Scientific Observations Required to Understand the Antarctic Ozone Hole
Platforms & Measurements
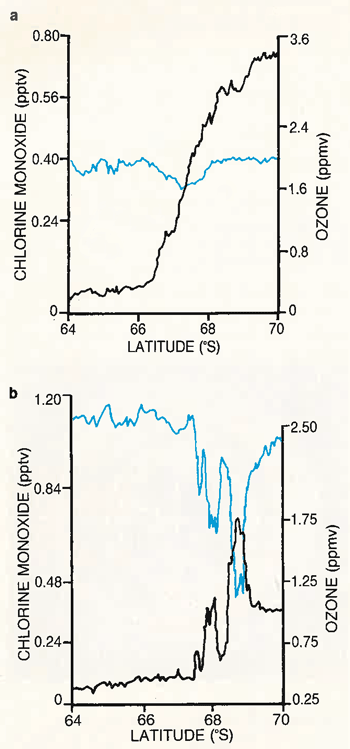
Figures: J.G. Anderson, W.H. Brune, M.J. Proffitt, Ozone destruction by chlorine radicals within the Antarctic vortex: The spatial and temporal evolution of ClO‐O3 anticorrelation based on in situ ER‐2 data, JGR-Atmospheres, doi:10.1029/JD094iD09p11465, 1989.
The NASA ER-2 high altitude research aircraft sampled air at those altitudes where the ozone hole was at its most intense, with data gathered on the air mass within the confines of the hole itself. The ER-2 instruments collected information on three-dimensional winds, pressure, temperature, temperature profiles +/- 1 km from flight level, chlorine monoxide, bromine monoxide, ozone, nitric oxide, reactive nitrogen, total water, nitrous oxide, whole air sampling, condensation nuclei, aerosol size distribution and composition, and cloud particle images and sizes.
The NASA DC-8 research aircraft flew at the lowermost extremities of the hole and deployed a combination of remote sounding of the overlying atmosphere with some in situ sampling. Vertical distributions of ozone and aerosols above the cruising altitude of the aircraft and within the hole were mapped. The DC-8 instruments collected ozone and aerosol profiles overhead by LIDAR; and measured ozone, bromine oxide, chlorine dioxide, nitrogen dioxide, nitric acid, and hydrogen chloride. In situ methods yielded ozone, total water, and whole air sampling.
Data Results
Results of the mission were presented at a Polar Ozone Symposium in Snowmass, Colorado in May of 1988. A two volume special issue of the Journal of Geophysical Research devoted to this experimental effort was published in August and November of 1989. The data obtained during the Antarctic mission show the lowest ozone levels ever recorded and directly implicate man-made chemical compounds, chlorofluorocarbons, in the enormous ozone loss over this remote region in the southern hemisphere. One of the most compelling pieces of evidence leading to this conclusion was the data collected 16 September 1987. These data, measured on the ER-2 aircraft as it flew south from Chile into the ozone hole, show the dramatic inverse correlation between ozone and chlorine monoxide. Because chlorine monoxide is produced by the process in which manmade chlorine destroys ozone, the large quantities observed provide strong evidence that manmade chemicals are involved in the Antarctic ozone loss process.