Scientific Assessment of Ozone Depletion: 1994
Common Questions About Ozone

1. How Can Chlorofluorocarbons (CFCs) Get to the Stratosphere If They're Heavier than Air?
Although the CFC molecules are indeed several times heavier than air, thousands of measurements have been made from balloons, aircraft, and satellites demonstrating that the CFCs are actually present in the stratosphere. The atmosphere is not stagnant. Winds mix the atmosphere to altitudes far above the top of the stratosphere much faster than molecules can settle according to their weight. Gases such as CFCs that are insoluble in water and relatively unreactive in the lower atmosphere (below about 10 km) are quickly mixed and therefore reach the stratosphere regardless of their weight.
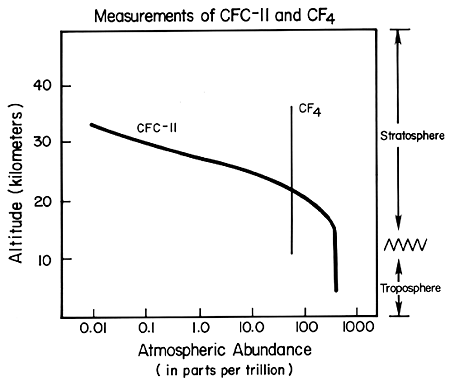
Much can be learned about the atmospheric fate of compounds from the measured changes in concentration versus altitude. For example, the two gases carbon tetrafluoride (CF4, produced mainly as a by-product of the manufacture of aluminum) and CFC-11 (CCl3F, used in a variety of human activities) are both much heavier than air. Carbon tetrafluoride is completely unreactive in the lower 99.9% of the atmosphere, and measurements show it to be nearly uniformly distributed throughout the atmosphere as shown in the figure. There have also been measurements over the past two decades of several other completely unreactive gases, one lighter than air (neon) and some heavier than air (argon, krypton), which show that they also mix upward uniformly through the stratosphere regardless of their weight, just as observed with carbon tetrafluoride. CFC-11 is unreactive in the lower atmosphere (below about 15 km) and is similarly uniformly mixed there, as shown. The abundance of CFC-11 decreases as the gas reaches higher altitudes, where it is broken down by high energy solar ultraviolet radiation. Chlorine released from this breakdown of CFC-11 and other CFCs remains in the stratosphere for several years, where it destroys many thousands of molecules of ozone.