Scientific Assessment of Ozone Depletion: 1994
Common Questions About Ozone

Ozone is exceedingly rare in our atmosphere, averaging about 3 molecules of ozone for every ten million air molecules. Nonetheless, atmospheric ozone plays vital roles that belie its small numbers. This Appendix to the World Meteorological Organization/United Nations Environment Programme (WMO/UNEP) Scientific Assessment of Ozone Depletion: 1994 answers some of the questions that are most commonly asked about ozone and the changes that have been occurring in recent years.
These common questions and their answers were discussed by the 80 scientists from 26 countries who participated in the Panel Review Meeting of the Scientific Assessment of Ozone Depletion: 1994. Therefore, this information is presented by a large group of experts from the international scientific community.
Ozone is mainly found in two regions of the Earth's atmosphere. Most ozone (about 90%) resides in a layer between approximately 10 and 50 kilometers (about 6 to 30 miles) above the Earth's surface, in the region of the atmosphere called the stratosphere. This stratospheric ozone is commonly known as the "ozone layer." The remaining ozone is in the lower region of the atmosphere, the troposphere, which extends from the Earth's surface up to about 10 kilometers. The figure below shows this distribution of ozone in the atmosphere.
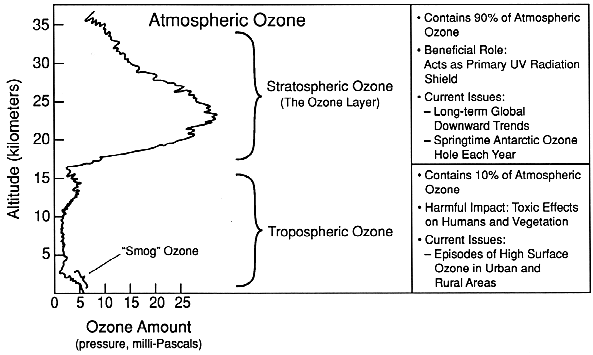
While the ozone in these two regions is chemically identical (both consist of three oxygen atoms and have the chemical formula "O3"), the ozone molecules have very different effects on humans and other living things depending upon their location.
Stratospheric ozone plays a beneficial role by absorbing most of the biologically damaging ultraviolet sunlight called UV-B, allowing only a small amount to reach the Earth's surface. The absorption of UV radiation by ozone creates a source of heat, which actually forms the stratosphere itself (a region in which the temperature rises as one goes to higher altitudes). Ozone thus plays a key role in the temperature structure of the Earth's atmosphere. Furthermore, without the filtering action of the ozone layer, more of the Sun's UV-B radiation would penetrate the atmosphere and would reach the Earth's surface in greater amounts. Many experimental studies of plants and animals, and clinical studies of humans, have shown the harmful effects of excessive exposure to UV-B radiation (these are discussed in the WMO/UNEP reports on impacts of ozone depletion, which are companion documents to the WMO/UNEP scientific assessments of ozone depletion).
At the planet's surface, ozone comes into direct contact with life-forms and displays its destructive side. Because ozone reacts strongly with other molecules, high levels are toxic to living systems and can severely damage the tissues of plants and animals. Many studies have documented the harmful effects of ozone on crop production, forest growth, and human health. The substantial negative effects of surface-level tropospheric ozone from this direct toxicity contrast with the benefits of the additional filtering of UV-B radiation that it provides.
With these dual aspects of ozone come two separate environmental issues, controlled by different forces in the atmosphere. In the troposphere, there is concern about increases in ozone. Low-lying ozone is a key component of smog, a familiar problem in the atmosphere of many cities around the world. Higher than usual amounts of surface-level ozone are now increasingly being observed in rural areas as well. However, the ground-level ozone concentrations in the smoggiest cities are very much smaller than the concentrations routinely found in the stratosphere.
There is widespread scientific and public interest and concern about losses of stratospheric ozone. Ground-based and satellite instruments have measured decreases in the amount of stratospheric ozone in our atmosphere. Over some parts of Antarctica, up to 60% of the total overhead amount of ozone (known as the "column ozone") is depleted during September and October. This phenomenon has come to be known as the Antarctic "ozone hole." Smaller, but still significant, stratospheric decreases have been seen at other, more-populated regions of the Earth. Increases in surface UV-B radiation have been observed in association with decreases in stratospheric ozone.
The scientific evidence, accumulated over more than two decades of study by the international research community, has shown that human-made chemicals are responsible for the observed depletions of the ozone layer over Antarctica and likely play a major role in global ozone losses. The ozone-depleting compounds contain various combinations of the chemical elements chlorine, fluorine, bromine, carbon, and hydrogen, and are often described by the general term halocarbons. The compounds that contain only carbon, chlorine, and fluorine are called chlorofluorocarbons, usually abbreviated as CFCs. CFCs, carbon tetrachloride, and methyl chloroform are important human-made ozone-depleting gases that have been used in many applications including refrigeration, air conditioning, foam blowing, cleaning of electronics components, and as solvents. Another important group of human-made halocarbons is the halons, which contain carbon, bromine, fluorine, and (in some cases) chlorine, and have been mainly used as fire extinguishants. Governments have decided to discontinue production of CFCs, halons, carbon tetrachloride, and methyl chloroform, and industry has developed more "ozone-friendly" substitutes.
Two responses are natural when a new problem has been identified: cure and prevention. When the problem is the destruction of the stratospheric ozone layer, the corresponding questions are: Can we repair the damage already done? How can we prevent further destruction? Remedies have been investigated that could (i) remove CFCs selectively from our atmosphere, (ii) intercept ozone-depleting chlorine before much depletion has taken place, or (iii) replace the ozone lost in the stratosphere (perhaps by shipping the ozone from cities that have too much smog or by making new ozone). Because ozone reacts strongly with other molecules, as noted above, it is too unstable to be made elsewhere (e.g., in the smog of cities) and transported to the stratosphere. When the huge volume of the Earth's atmosphere and the magnitude of global stratospheric ozone depletion are carefully considered, approaches to cures quickly become much too expensive, impractical, and potentially damaging to the global environment. Prevention involves the internationally agreed-upon Montreal Protocol and its Amendments and Adjustments, which call for elimination of the production and use of the CFCs and other ozone-damaging compounds within the next few years. As a result, the ozone layer is expected to recover over the next fifty years or so as the atmospheric concentrations of CFCs and other ozone-depleting compounds slowly decay.
The current understanding of ozone depletion and its relation to humankind is discussed in detail by the leading scientists in the world's ozone research community in the Scientific Assessment of Ozone Depletion: 1994. The answers to the common questions posed below are based upon that understanding and on the information given in earlier WMO/UNEP reports.